Modi takes homegrown COVID jab as India expands vaccination drive
Indian PM urges every eligible person to get vaccine in an effort to rid the country of the new coronavirus.
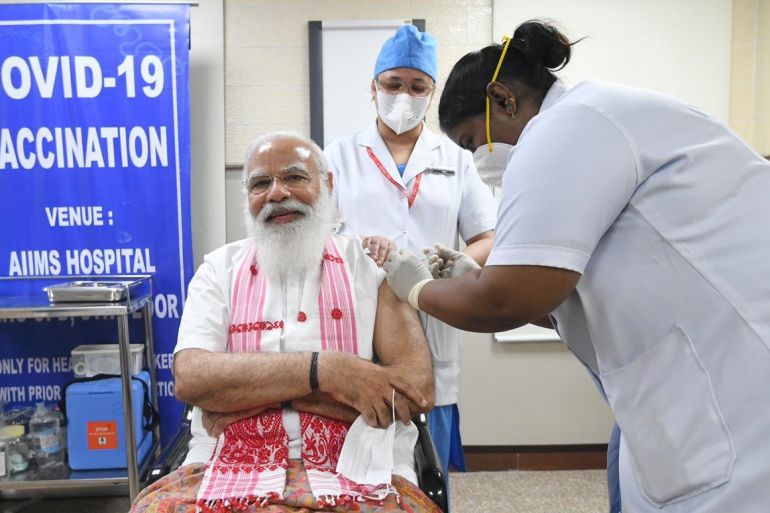
India’s prime minister has tweeted a photo of himself receiving a first dose of a homegrown COVID-19 vaccine, kicking off an expansion of the country’s immunisation campaign that began in mid-January with healthcare workers.
The 70-year-old Prime Minister Narendra Modi, who took his mask off to smile for the photo, said in a tweet on Monday that he urges every eligible person to get the vaccine in an effort to rid India of the new coronavirus.
Keep reading
list of 3 itemsIndia tightens regulatory grip on social media companies
India, China FMs to set up hotline as border crisis eases
“Took my first dose of the COVID-19 vaccine at AIIMS,” Modi tweeted, referring to the All India Institute of Medical Sciences hospital in the capital, New Delhi.
“I appeal to all those who are eligible to take the vaccine. Together, let us make India COVID-19 free,” the Indian leader added.
Took my first dose of the COVID-19 vaccine at AIIMS.
Remarkable how our doctors and scientists have worked in quick time to strengthen the global fight against COVID-19.
I appeal to all those who are eligible to take the vaccine. Together, let us make India COVID-19 free! pic.twitter.com/5z5cvAoMrv
— Narendra Modi (@narendramodi) March 1, 2021
India, which has reported the highest number of COVID-19 cases in the world after the United States, has so far vaccinated more than 12 million health and front-line workers.
The country is now expanding its vaccination drive beyond healthcare and front-line workers, offering the shots to older people and those with medical conditions that put them at risk.
Those now eligible to be vaccinated include people over 60, as well as those over 45 who have ailments such as heart disease or diabetes that make coronavirus infections more dangerous.
The shots will be given for free at government hospitals and will also be sold at over 10,000 private hospitals at a fixed price of 250 rupees, or $3.40, per shot.
The government said last week it would let people choose their vaccination centres, effectively letting beneficiaries pick either the homegrown COVAXIN shot or the AstraZeneca vaccine, unlike earlier.
The inoculation campaign has progressed slower than expected due to a reluctance of health and front-line workers to take COVAXIN, which was approved without late-stage efficacy data.
Only about 11 percent of vaccinated people have opted for the product developed by Bharat Biotech and the state-run Indian Council of Medical Research.
Bharat Biotech has said efficacy data from a late-stage trial on nearly 26,000 volunteers who took COVAXIN will be out soon.
The company, along with India’s drug regulator, says COVAXIN is safe and effective, based on early and intermediate studies.
India has reported more than 11 million coronavirus infections and over 157,000 deaths.